Shells and subshells
All the orbitals of a given value of n are said to form a single shell of the atom. In a hydrogenic atom, all orbitals of given n, and therefore belonging to the same shell, have the same energy. It is common to refer to successive shells by letters:
n=1 2 3 4...
K L M N...
Thus, all the orbitals of the shell with n = 2 form the L shell of the atom, and so on. The orbitals with the same value of n but different values of l are said to form a sub shell of a given shell. These subshells are generally referred to by letters:
l = 0 1 2 3 4 5 6...
s p d f g h i...
The letters then run alphabetically (j is not used). Figure 10.7 is a version of Fig. 10.5 which shows the subshells explicitly. Because l can range from 0 to n − 1, giving n values in all, it follows that there are n subshells of a shell with principal quantum numbern. Thus, when n =1, there is only one subshell, the one with l = 0. When n = 2, there are two subshells, the 2s subshell (with l = 0) and the 2p subshell (with l = 1). When n = 1 there is only one subshell, that with l = 0, and that subshell contains only one orbital, with ml = 0 (the only value of ml permitted). When n = 2, there are four orbitals, one in the s subshell with l = 0 and ml = 0, and three in the l = 1 subshell with ml =+1, 0, −1. When n = 3 there are nine orbitals (one with l = 0, three with l = 1, and five with l = 2). The organization of orbitals in the shells is summarized in Fig. 10.8. In general, the number of orbitals in a shell of principal quantum number n is n2, so in a hydrogenic atom each energy level is n2-fold degenerate.
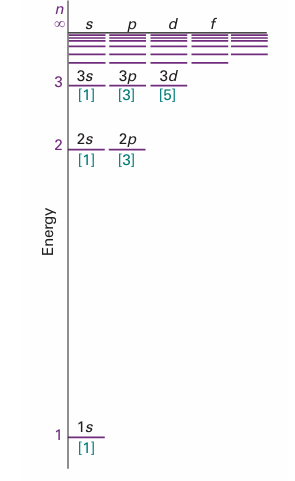
Fig. 10.7 The energy levels of the hydrogen atom showing the subshells and (in square brackets) the numbers of orbitals in each subshell. In hydrogenic atoms, all orbitals of a given shell have the same energy.
